Stability of Tea Polyphenol (−)-Epigallocatechin-3-gallate and Formation of Dimers and Epimers under Common Experimental Conditions | Journal of Agricultural and Food Chemistry
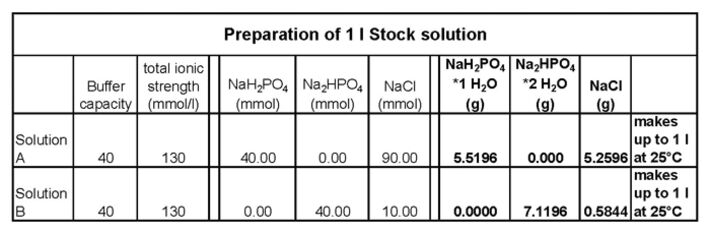
Puffer 0.1 M NaOH sowie 0.1 M HCl (Referrenz) NaHCO3 mit Na2CO3 (Puffer I, wird blau, pH ca. 9) NaH2PO4 mit Na2HPO4 (Puff
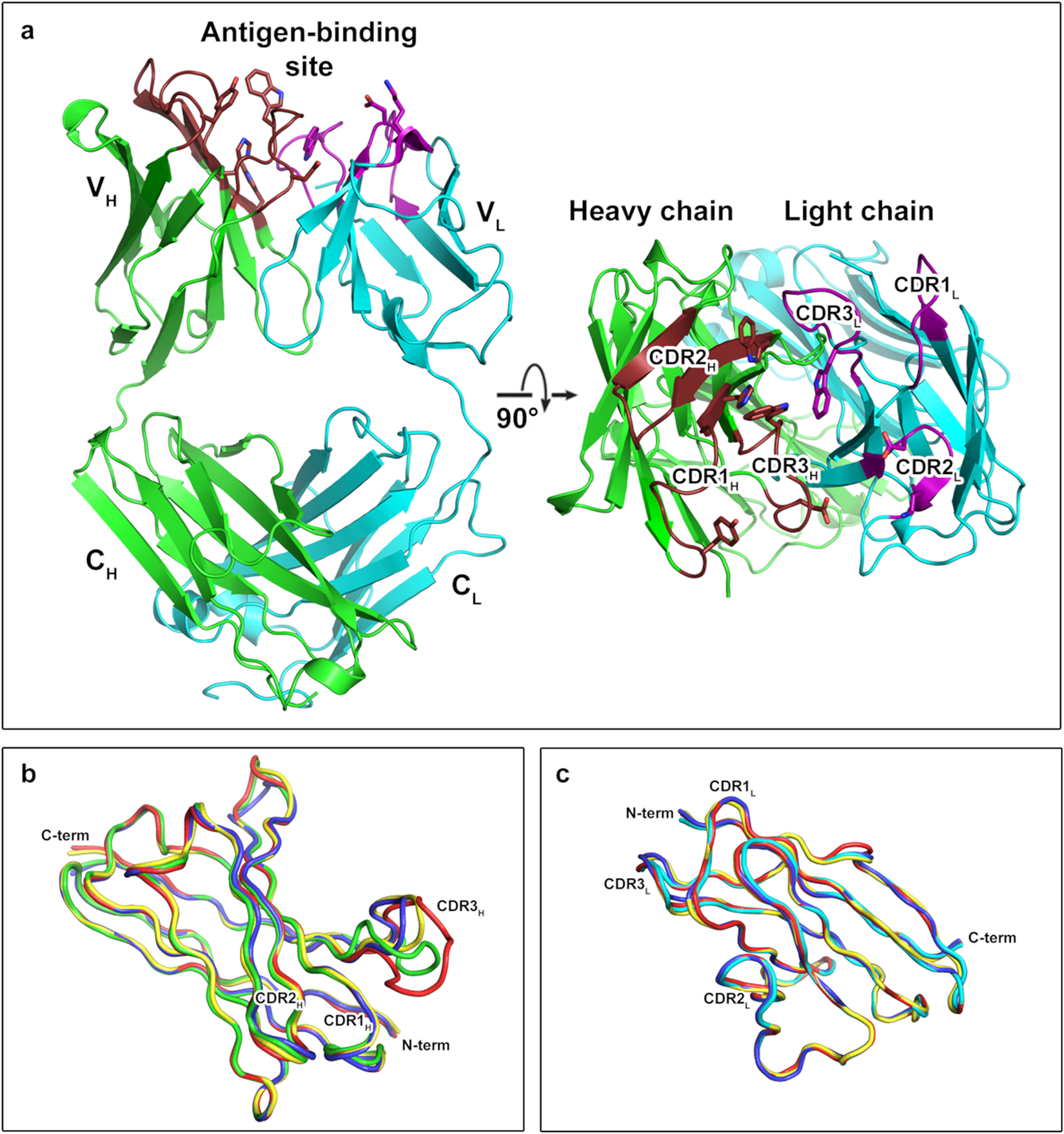
Structural insights into antigen recognition of an anti-β-(1,6)-β-(1,3)-D-glucan antibody | Scientific Reports

Thermodynamic and Kinetic Analysis of Isothermal Titration Calorimetry Experiments by Using KinITC in AFFINImeter | SpringerLink

Toxins | Free Full-Text | Tetrodotoxin/Saxitoxins Selectivity of the Euryhaline Freshwater Pufferfish Dichotomyctere fluviatilis | HTML

Synergetic Determination of Thermodynamic and Kinetic Signatures Using Isothermal Titration Calorimetry: A Full-Curve-Fitting Approach | Analytical Chemistry

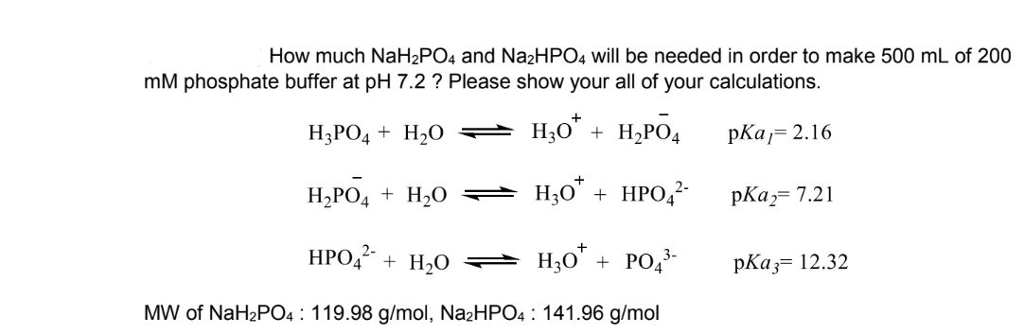
SOLVED: A phosphate buffer was made using the sodium salts of NaH2PO4 and Na2HPO4. The pH was recorded as 8.3 but you desire a pH of 7.5. To establish the desired pH,